 
Because the fuel in a reactor is only three to four percent U-235, the reactor cannot explode like a nuclear bomb. However, to produce a large explosion, the uranium needs to be enriched to more than 50 percent U-235. This is the basics behind a nuclear weapon. If enough U-235 nuclei are close together, the process can accelerate, producing a lot of energy in a short time. Therefore, if a single U-235 nucleus fissions, the emitted neutrons can induce a fission in two or more U-235 nuclei, which each in turn can produce two or more, etc. However, U-235 can fission if a neutron hits it. The rate of U-235 spontaneous fission is very slow, too slow to be of any use in a nuclear reactor. In fact, radioactive decay of fission products produces about 7% of the heat in a nuclear reactor that has been operating for several months. Because the radiation has energy, it is also a source of heat in a nuclear reactor. Collectively, the beta particle and gamma rays are called radiation and this radiation has energy. In addition, when a fission product decays, the nucleus emits one or more gamma rays. A radioactive nucleus changes (decays) to another nucleus by emitting a particle for fission products, this is a beta particle, which is just a high speed electron. One aspect of the fission products, which was a major factor in the TMI accident, is that the fission products are radioactive. To have U-235 fission efficiently, the uranium fuel in a reactor is enriched the uranium goes through a process to increase the content of U-235 from 0.7% to 3 to 4%. The fission products and the neutrons have energy, which is the source of heat. The most important difference is that U-235 spontaneously splits, or fissions, producing two smaller nuclei (called fission products) plus two to five neutrons. Even though both U-238 and U-235 are uranium, they have different characteristics. However, not all uranium atoms have 146 neutrons 0.7% have 143, and these are called U-235. We call this number the mass number, and refer to these atoms as uranium-238, or just U-238. Most uranium atoms (99.3%) have a nucleus with 146 uncharged neutrons and 92 positively charged protons, for a total of 238 neutrons and protons. The fuel for a nuclear reactor is uranium, but not just any uranium. 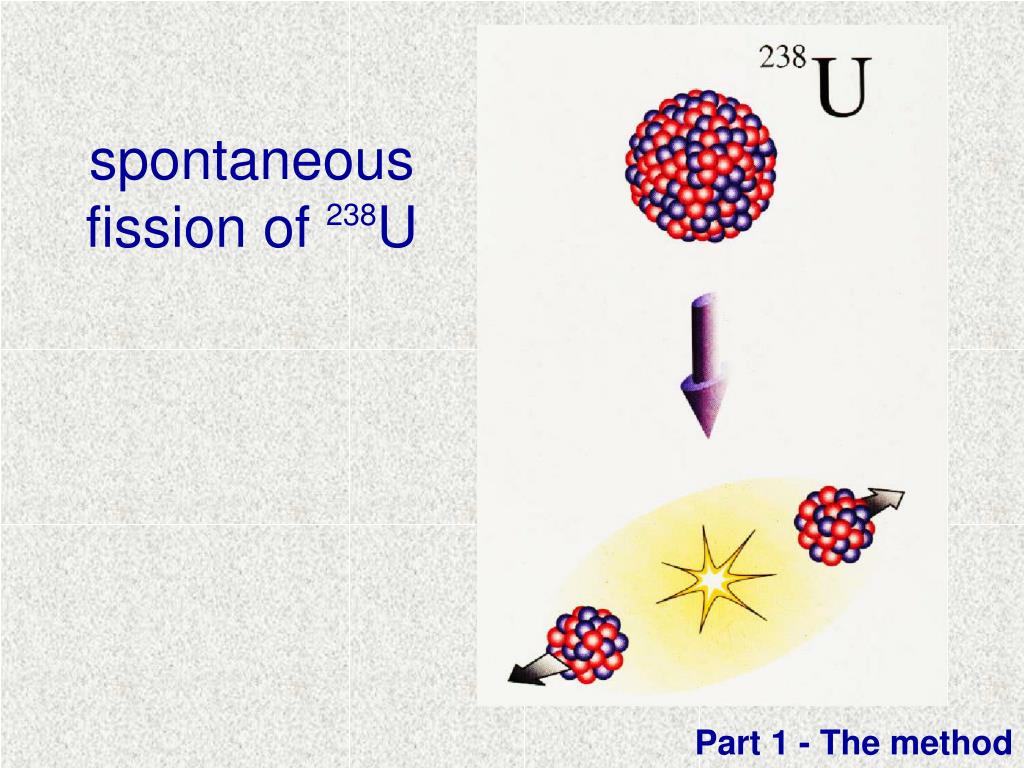
Although the basic process is simple, making it work is rather complicated.įirst, let’s look at the process of how nuclear fuel produces heat. In a nuclear plant the source of heat is a nuclear reactor. In fossil fuel plants the source of heat is burning coal, oil, or gas. The steam then turns a turbine, the turbine turns an electrical generator, and the generator produces electricity. To produce electricity, a power plant needs a source of heat to boil water which becomes steam. The only purpose of a nuclear power plant is to produce electricity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
